- Cet évènement est passé.
Séminaire Diane Rebiscoul (CEA ICSM)

The role of nanoconfinement on water properties and specific ion
effects on the evolution of porous silica
Processes taken place at the solid/aqueous solution interface have a strong impact on
the evolution of materials in the fields of construction, environment, geochemistry, membranes,
catalysis, and nuclear wastes. Since many materials in these application fields are completely
or partially nanoporous (cementitious materials, biominerals, clay, secondary minerals…) and
filled with aqueous solutions, the processes and chemical reactions occurring in this
nanoconfinement has a major impact on the materials evolution on a macroscopic scale. The
study of such processes and chemical reactions occurring in nanometer-sized porosities in
contact with aqueous solutions (ions sorption, electrolyte diffusion, phase precipitation, pore
wall dissolution and the recondensation of dissolved species) is important since it differs from
the ones occurring in dense materials. Since water takes part in most of these processes and
chemical reactions, the understanding of water molecules behavior in such media is essential to
be able to predict the behavior of such materials.
Generally, the macroscopic evolution of materials in solution is described or predicted
using modelling. Thermodynamic models and rate laws developed and integrated in computer
programs (PHREEQC, JChess…) generally take into account some data arising from
measurements performed in diluted media (concentration of dissolved ions, pH, kinetics and
thermodynamic constants…). For nanoporous materials, the validity of these models and rate
laws are still not proven yet, thus prediction can be defective. Indeed, under confinement and
in presence of ions, the water behavior is modified by the strong interactions between the pore
surfaces and the ions structuring water, and slowing down its dynamics/transport from
nanoscale to macro-scale. Such effects modify the electrostatic interactions in the system and
thus the ions free energy landscape. Since chemical reactions such as silica
hydrolysis/recondensation processes are controlled by the solution-silica interfacial layer, the
study of the modification of this interfacial layer in nanoconfinement is of paramount
importance to describe the evolution of nanoporous materials in aqueous solution. In-fine, this
will allow the improvement of the thermodynamic models and rate laws of processes occurring
in nanoconfinement.
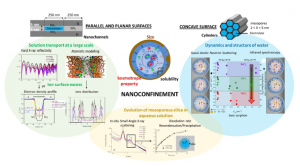
In this study, we investigated the water properties (structure and dynamics) in the
presence of ions in silica nanoconfinement and relate it to the evolution of silica mesoporous
materials in aqueous solutions. We used an original approach, consisting in the use of
electrolyte solutions having ions with various kosmotropic property XCl2 (X = Ba, Ca, Mg)
confined in model systems such as two parallel and plane silica surfaces spaced of 3 and 5 nm
(nanochannels) and highly ordered mesoporous silica materials represented by SBA-15 (6 nm
pore size and microporous pore wall) and MCM-41 (3 nm pore size and dense pore wall).