Controlled Porosity materials
Scientific objectives
The team is renowned worldwide for its activity in synthesis and study of porous and nanostructured materials for applications in the fields of adsorption, heterogeneous catalysis, ion exchange, environmental protection and energy storage.
Our research work aims to :
- Develop synthetic strategies thanks to the use of novel and/or biosourced structuring agents for the elaboration of new structures in families of zeolites and related compounds as well as coordination polymers (Metal-Organic Frameworks)
- Develop or optimise synthetic processes for inorganic and hybrid 3D materials by soft chemistry and hydrothermal routes
- Understand properties through detailed characterizations
- Study the storage of mechanical energy by intrusion-extrusion experiments of non-wetting liquids in porous materials under high pressure.
Members

Magali BONNE
Assistant professor
➜ CV

Gérald CHAPLAIS
Assistant professor
Co-leader
➜ CV

Jean DAOU
Professor
➜ CV

Bénédicte LEBEAU
Principal investigator
Co-leader
➜ CV

Claire MARICHAL
Professor
➜ CV

Habiba NOUALI
Research engineer
➜ CV

Jean-Louis PAILLAUD
Principal investigator
➜ CV

Emmanuel OHEIX
Assistant professor
➜ CV

Taylan ORS
Assistant professor
➜ CV

Severinne RIGOLET
Research engineer
➜ CV

Andrey RYZHIKOV
Research scientist
➜ CV

Angélique SIMON-MASSERON
Professor
➜ CV
Non-permanent members
CHAIB DRAA
Yacine Malik
Doctorant
➜ CV
WIDOLFF
Marina
Doctorante
➜ CV
FROEHLY
Marie
Doctorante
➜ CV
GUICHENEY
Grégory
Post-Doctorant
➜ CV
PERNET
Samuel
Doctorant
➜ CV
PREMET
Laura
Ingénieure
➜ CV
SEBBAT
Malik
Doctorant
➜ CV
Research fields

Elaboration of new 3D porous materials, development of new/optimized synthesis processes, functionalization (post-synthesis or direct synthesis) and shaping (direct or ex situ)
M. Bonne, G. Chaplais, J. Daou, B. Lebeau, J.-L. Paillaud, J. Patarin, A. Ryzhikov, A. Simon-Masseron
Synthesis of microporous germanosilicates of topology -CLO and UTL. A new family of pyrrolidine derivatives was synthesized and tested in hydrothermal synthesis of microporous germanosilicates as organic structure directing agents (OSDA). One of them, N,N-dimethylperhydro-dicyclopenta[b, d]pyrrolium, made it possible to obtain a germanosilicate of topology -CLO with a very high purity and a Si/Ge ratio of 2, an absolute record for this topology initially known as gallophosphate. Similarly, (6R, 10S)-6,10-dimethyl-5-azoniaspiro [4,5]decane led to IM-12 of topology UTL, a microporous germanosilicate.

Brevet
M. Dodin, J. -L. Paillaud, P. Caullet, A. Defoin, Patent WO2013064786A1, 2013

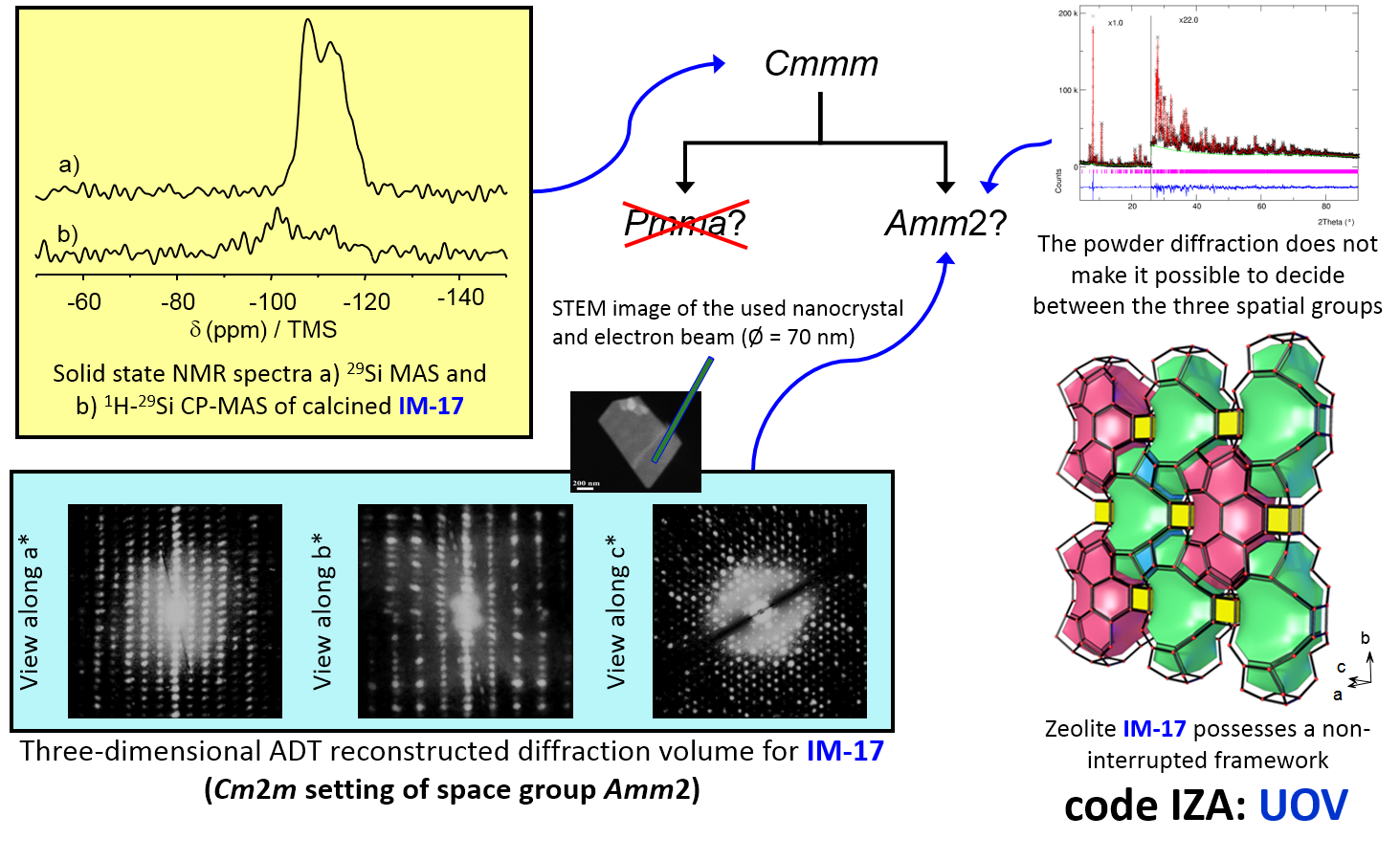
Multiscale structural characterization of porous solids by coupling of powder X- rays diffraction and solid-state NMR
M. Bonne, G. Chaplais, J. Daou, L. Josien, B. Lebeau, C. Marichal, L. Michelin, J.-L. Paillaud, J. Patarin, A. Ryzhikov, S. Rigolet, A. Simon-Masseron
29Si MAS NMR and 1H-29Si CP/MAS NMR were used to distinguish between two possible structures of zeolite IM-17 initially described in the Cmmm space group, one of which is interrupted. In addition, recent techniques of nano-electron diffraction have also confirmed the choice of the non-centrosymmetric space subgroup Amm2 (collaboration with Johannes Gutenberg, University Mainz).
Publication
Y. Lorgouilloux, M. Dodin, E. Mugnaioli, C. Marichal, P. Caullet, N. Bats, U. Kolb, U. J.-L. Paillaud RSC Advances 2014, 4, 19440 DOI : 10.1039/c4ra01383b

Study of the formation mechanisms of porous solids
G. Chaplais, J. Daou, B. Lebeau, C. Marichal, J.-L. Paillaud, J. Patarin, S. Rigolet, A. Simon-Masseron
Thanks to the support of molecular modelling, various organic compounds capable of acting both as OSDAs and inhibitors in the crystal growth of materials (syntheses of nano-sponges or zeolite nanosheets) have been identified. This approach enables the preparation of ZSM-5 zeolite nanosheets by structural conversion of an in situ formed lamellar polysilicate magadiite in the presence of a mono-nitrogen bifunctional compound. This approach was then applied to the synthesis of zeolites EMC-1 (FAU) and EMC-2 (EMT) in the presence of a new bifunctional OSDA of di-aza-crown ether-type.

Publication
J. Dhainaut, T. J. Daou, Y. Bidal, N. Bats, B. Harbuzaru, G. Lapisardi, H. Chaumeil, A. Defoin, L. Rouleau, J. Patarin CrystEngComm 2013, 15, 3009 DOI 10.1039/C3CE40118A

Optimization of materials and their properties through in-depth structural characterizations
M. Bonne, G. Chaplais, J. Daou, L. Josien, B. Lebeau, C. Marichal, L. Michelin, H. Nouali, J.-L. Paillaud, J. Patarin, A. Ryzhikov, S. Rigolet, A. Simon-Masseron
It has been shown for the first time that the endogenous radicals present in thermally carbonized porous silicas (TCPSi) can be used to improve the sensitivity of 1H-13C CPMAS NMR using dynamic nuclear polarization (DNP). The originality of this work lies in the fact that a significant improvement in the 13C NMR signal of the TCPSi material was obtained without the addition of polarization agents. The presence of stable radicals in the TCPSi material has been demonstrated and their quantity determined by Electron Paramagnetic Resonance (EPR). The feasibility of the 1H-13C CPMAS DNP NMR experiments has been proven and this technique has been used to obtain information on the chemical nature of the TCPSi.

Publication
J. Riikonen, S. Rigolet, C. Marichal, F. Aussenac, J. Lalevée, F. Morlet-Savary, P. Fioux, C. Dietlin, M. Bonne, B. Lebeau, V.-P. Lehto, Journal of Physical Chemistry C 2015, 119, 19272 DOI : 10.1021/acs.jpcc.5b05970

Evaluation of the adsorption capacity of pollutants in the gas phase and in the liquid phase by porous materials
G. Chaplais, J. Daou, B. Lebeau, H. Nouali, J. Patarin, A. Simon-Masseron
The syntheses of conventional zeolites result in powdered products, which are generally penalizing for numerous industrial applications. Therefore, the development of shaping methods for these materials with controllable morphologies and sizes is of great technological importance. In the case of the ADEME Méterdiox+ project, it is expected that the field device will be equipped with cartridges into which dioxins/furans adsorbent materials will be introduced. Consequently, the latter must be shaped to avoid the loss of load and the clogging of the cartridges observed when powders are used, as much as possible.

Publication
G. Rioland, L. Bullot, T. J. Daou, A. Simon-Masseron, G. Chaplais, D. Faye, E. Fiani, J. Patarin RSC Advances 2016, 6, 2470 DOI : 10.1039/c5ra23258a

Development of efficient systems for the storage or the absorption of mechanical energy based on hydrophobic porous solids
G. Chaplais, J. Daou, B. Lebeau, C. Marichal, H. Nouali, J.-L. Paillaud, J. Patarin, A. Ryzhikov
The water intrusion-extrusion in hydrophobic pure silica zeolites (zeosils) leads to a molecular spring behaviour in the case of Silicalite-1 zeolite of topology MFI. During the extrusion, almost complete restitution of the stored energy is observed (energetic yield of 98 %). When concentrated solutions of electrolytes, such as LiCl, MgCl2 or NaCl, are used instead of pure water, the energetic performances of the zeosils increase considerably. Thus, the stored energy is tripled in the case of the “Silicalite-1 – LiCl 20 M” system compared to one for the “Silicalite-1 – water” system.

Publication
I. Khay, T. J. Daou, H. Nouali, A. Ryzhikov, S. Rigolet, J. Patarin Journal of Physical Chemistry C 2014, 118, 3935 DOI : 10.1021/jp4105163

Specific facilities

ZETASIZER
Zeta potential and particles size measurements with DLS
Equipment : ZETASIZER Nano Series Nano-Zs (Malvern Instruments)
Contact : J. Daou

Mercury Porosimetry
Micromeritic’s Autopore IV 9500 series (200 MPa, 400 MPa)
Contact : H. Nouali

VOCs emissions test chamber
Equipment : Climtech, V = 50 L
Contact : H. Nouali

Thermogravimetric equipment
Modified thermogravimetric equipment (kinetic measurements and adsorption isotherms of VOCs, water vapor….) coupled with a mass spectrometer
Equipment : SETSYS Vapor Sorption (Setaram), QGA (Hiden Analytical)
Contact : H. Nouali / J. Daou
Gas chromatography with mass spectrometer and thermodesorption system
Equipment : TurboMatrix 300 (PerkinElmer) ; CP3800 (Varian) ; Saturn 2200 (Varian)
Contact : H. Nouali

Mixer
Equipment : EL1 (Eirich)
Contact : J. Daou

Atomic absorption instrument
Equipment : AA 240 FS (Varian)
Contact : L. Michelin

Autoclaves
Equipment : design IUT Mulhouse, Top Industrie, Autoclave France, Parr. V = 2 to 750 mL
Contact : J-L. Paillaud
Main collaborators
Laboratoire de Photochimie et Ingénierie Moléculaire (UHA, Mulhouse, France)
Laboratoire de Gestion des Risques et Environnement (UHA, Mulhouse, France)
Laboratoire d’Innovation Moléculaire et Applications (UHA-UNISTRA, Mulhouse, France)
Laboratoire de Physique et Mécanique Textiles (UHA, Mulhouse, France)
Institut de Physique et de Chimie des Matériaux de Strasbourg (UNISTRA, Strasbourg, France)
Laboratoire de Chimie de Coordination (UNISTRA, Strasbourg, France)
Institut de Chimie de Strasbourg, Equipe Synthèse et réactivité organiques et Catalyse (UNISTRA, Strasbourg, France)
Laboratoire Structure et Réactivité des Systèmes Moléculaires Complexes (Univ. Lorraine, Vandoeuvre-lès-Nancy, France)
Institut Carnot de Bourgogne (Univ. Bourgogne, Dijon, France)
Institut de Recherche de Chimie de Paris (Chimie ParisTech, Paris, France)
Laboratoire Processus d’Activation Sélective par Transfert d’Energie Uni-Electronique ou Radiatif (Sorbonne Université, Paris, France)
Laboratoire de Matériaux Divisés, Interfaces, Réactivité, Electrochimie (Univ. Aix-Marseille, Marseille, France)
Institut de Chimie des Milieux et Matériaux de Poitiers (Univ. Poitiers, Poitiers, France)
Department of Geology, Mineralogy and Geophysics (Univ. Bochum, Germany)
Laboratory of Materials, Catalysis, Environment and Analytical Methods Faculty of Sciences (Univ. Libanaise, Beyrouth, Lebanon)
Laboratoire de Chimie des Matériaux (Univ. Es Sénia, Oran, Algeria)